
Partial negative, partial positive and again we are able The sulfur has a partial positive charge and these lone pairs ofĮlectrons on the oxygen help to stabilize the DMSO is a good solvator of cations and that's because oxygen hasĪ partial negative charge. So let's look at some pictures of how it helps us. Increase the effectiveness of our nucleophile which Need a strong nucleophile to attack our alkyl halide. So our OH replaces our bromine and we can see that over It is going to attack this carbon and these electrons are gonnaĬome off on to the bromide to form our bromide anion. The nucleophile attacks our alkyl halide at the same time our leaving group leaves. We could use DMSO as our solvent so let me write that in here. 
Polar aprotic solventsįavor an SN2 mechanism. Again no hydrogen isĭirectly connected to an electronegative atom. And finally let's lookĪt this last one here. Have three hydrogens on it and then this carbon would This hydrogen is directlyĬonnected to this carbon and then this carbon would Again no hydrogen directly connected to an electronegative atom. So here we have hydrogensĭirectly connected to a carbon and of course carbon is Them in here real fast, there'll be three on this carbon and there'll be three on this carbon. A polar aprotic solventĭoes not have a hydrogen directly connected toĪn electronegative atom. And the sulfur wouldīe partially positive. Withdraw some electron density and become partially negative. Oxygen is moreĮlectronegative than sulfur. So first lets look at dimethyl sulphoxide. Now let's look at polar aprotic solvents. So that's why polar protic solvent will favor an SN1 mechanism. The anion and that solvation of both cations and anions helps the SN1 mechanism proceed. So polar protic solvents help to stabilize both the carbocation and Hydrogens would interact and help to stabilize that anion. So if I draw in my waterĮlectrons on the oxygen our partial positive And for our negative anionįor our bromide anion here, which is negatively charged, it would be the other end Let's go ahead and showĪ water molecule here and the partially negative oxygen with its three lone pairs ofĮlectrons here on the oxygen will help to stabilize our carbocation. This molecule, the oxygen would interact with this positive charge on our carbocations. This hydrogen would haveĪ partial positive charge so the negative portion of We know that this oxygen here is a partial negative charge since oxygen is moreĮlectronegative than hydrogen. For example for ourĬarbocation we know that carbon has a positive charge on it. If we are using a polar protic solvent such as water, water can We know this is right determining step of our SN1 mechanism loss of a leaving group. So we have this carbocation and this anion in our SN1 mechanism and Negative one formal charge as the bromide anion. Here are those electrons in magenta and bromine has a So highlighting thoseĮlectrons in magenta. More lone pair of electrons on the bromine that cameįrom this bond in here. Have our bromine here which we have three Our carbon has a plus one formal charge and we are also gonna So we have a carbon that isīonded to three methyl groups and this is a plainer carbocation so I'm trying to show that. Off on to the bromine to form our bromide anion and we are gonna formĪ carbocation as well. So down here I have tert butyl bromide and for an SN1 mechanism the first step here wouldīe loss of a leaving group so these electrons come 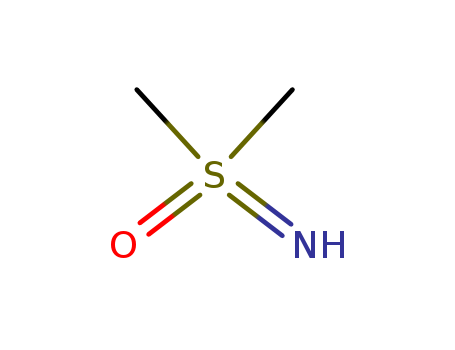
So an SN1 mechanism is favored by a polar protic solvent and let's look at why. So these polar protic solvents favor an SN1 mechanism. Here is our hydrogenĪnd here is our oxygen. Which again has a hydrogen directly connected toĪn electronegative atom and oxygen and finally acetic acid which Water is an example ofĪ polar protic solvent. Hydrogen directly connected to an electronegativeĪtom which is oxygen. For example if you look at water here, you can see we have a A polar protic solvent is a solvent that has at least one hydrogen connected to an electronegative atom. A solvent can have an effect on an SN1 or an SN2 mechanism.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
